Randomized controlled trial of levetiracetam versus fosphenytoin for convulsive status epilepticus in children
Abstract
Introduction: Convulsive status epilepticus (CSE) is a common neurological emergency in childhood carrying a risk of significant morbidity and mortality. The current recommended first choice second line treatment in children is phenytoin / fosphenytoin although there is lack of evidence for its use and it is associated with significant side effects. Emerging evidence suggest that intravenous (IV) levetiracetam may be more effective with fewer side effects. The objective of this study was to compare the efficacy and safety of IV levetiracetam as a second line treatment for CSE with IV fosphenytoin in Indian children.
Methods: Design: Prospective randomized controlled trial.
Study setting: Paediatric emergency department of a Medical College in Tamilnadu, India from January 2017 to June 2017.
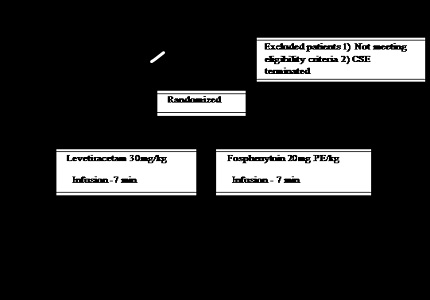
Study population: 50 children aged 3 months to 12 years admitted for CSE and in whom seizure has failed to terminate with 2 doses of benzodiazepine.
Intervention: 20mg Phenytoin equivalent (PE)/kg of fosphenytoin or 30mg/kg of levetiracetam administered intravenously over 7 minutes. Primary outcome: clinical cessation of seizures five minutes following the completion of the infusion of the study medication. Secondary outcome: seizure recurrence within 24 hours, drug related adverse events and length of PICU and hospital stay.
Results: Fosphenytoin terminated seizures earlier than levitiracetam (p= 0.029). There was no significant difference between the two drugs in other parameters including seizure control rate (p=0.667), seizure recurrence (p=0.44), seizure free duration (p=0.8), PICU stay (p=0.105) and hospital stay (p=0.311).
Conclusion: Levetiracetam may be an effective alternative to fosphenytoin in the management of convulsive status epilepticus in children.
Downloads
References
2. Chin RF, Neville BG,Peckham C, Bedford H,Wade A, Scott RC; NLSTEPSS Collaborative Group. Incidence, cause, and short-term outcome of convulsive status epilepticus in childhood: prospective population-based study. Lancet.2006 Jul 15;368(9531):222-9. [PubMed]
3. Raspall-Chaure M,Chin RF,Neville BG,Scott RC. Outcome of paediatric convulsive status epilepticus: a systematic review.Lancet Neurol.2006 Sep;5(9):769-79. [PubMed]
4. Treatment of convulsive status epilepticus. Recommendations of the Epilepsy Foundation of America's Working Group on Status Epilepticus.JAMA.1993 Aug 18;270(7):854-9.
5. Guidelines for epidemiologic studies on epilepsy. Commission on Epidemiology and Prognosis, International League Against Epilepsy.Epilepsia.1993 Jul-Aug;34(4):592-6. [PubMed]
6. Lowenstein DH,Bleck T,Macdonald RL. It's time to revise the definition of status epilepticus.Epilepsia.1999 Jan;40(1):120-2. [PubMed]
7. Appleton R,Macleod S,Martland T. Drug management for acute tonic-clonic convulsions including convulsive status epilepticus in children. Cochrane Database Syst Rev.2008 Jul 16;(3):CD001905. doi:10.1002/14651858.CD001905. [PubMed]
8. GlauserT, ShinnarS, GlossD, et al.,Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society.Epilepsy Curr. 2016 Jan-Feb; 16(1): 48–61.doi: 10.5698/1535-7597-16.1.48.
9. Reiter PD,Huff AD,Knupp KG,Valuck RJ. Intravenous levetiracetam in the management of acute seizures in children.Pediatr Neurol.2010 Aug;43(2):117-21. doi: 10.1016/j.pediatrneurol.2010.03.017. [PubMed]
10. Lynch BA, Lambeng N, Nocka K, Kensel-Hammes P, Bajjalieh SM, Matagne A, Fuks B. The synaptic vesicle protein SV2A is the binding site for the antiepileptic drug levetiracetam.Proc Natl Acad Sci U S A.2004 Jun 29;101(26):9861-6. Epub 2004 Jun 21. [PubMed]
12. Rogawski, MA. Diverse mechanisms of antiepileptic drugs in the development pipeline Epilepsy Res 2006 Jun;. 69(3): 273 -94. doi:10.1016/j.eplepsyres.2006.02.004. [PubMed]
13. Yasiry Z, Shorvon SD. The relative effectiveness of five antiepileptic drugs in treatment of benzodiazepine resistant convulsive status epilepticus : a meta – analysis of published studies. Seizure.2014Mar;23(3):167-74.doi:10.1016/j.seizure2013.12.007. [PubMed]
14. Jaclyn OC, Philip L, Wilson, Saely S. Levetiracetam is as effective as phenytoin in status epilepticus with less adverse events in the ED. Critical care medicine. 2015Dec; 43(12):6.doi:10. 1097/01.ccm. 0000473848.40708.ea.
15. Chakravarthi S,GoyalMK,Modi M,Bhalla A,Singh P. Levetiracetam versus phenytoin in management of status epilepticus.J Clin Neurosci.2015 Jun;22(6):959-63. doi: 10.1016/j.jocn.2014.12.013. Epub 2015 Apr 18. [PubMed]
16. Egunsola O,Choonara I,Sammons HM. Safety of Levetiracetam in Paediatrics: A Systematic Review.PLoS One.2016 Mar 1;11(3):e0149686. doi: 10.1371/journal.pone.0149686. eCollection 2016. [PubMed]
17. Browne TR. Fosphenytoin (Cerebyx).Clin Neuropharmacol.1997 Feb;20(1):1-12.
18. Ramsay RE,Wilder BJ,Uthman BM,Garnett WR,Pellock JM,Barkley GL,Leppik IE,Knapp LE. Intramuscular fosphenytoin (Cerebyx) in patients requiring a loading dose of phenytoin.Epilepsy Res.1997 Oct;28(3):181-7. [PubMed]
Copyright (c) 2018 Author (s). Published by Siddharth Health Research and Social Welfare Society

This work is licensed under a Creative Commons Attribution 4.0 International License.


 OAI - Open Archives Initiative
OAI - Open Archives Initiative


