Outcome of infants of HIV positive mothers following administration of nevirapine drops at secondary level referral centre
Abstract
Introduction: Prevention of transmission of HIV to new born and infants through the integration of preventive and care measures.
Objective: To assess HIV transmission, adverse reactions and morbidity among neonates and infants born to HIV positive mothers till 18months of age over a period of 10 years by prevention of mother to child transmission of HIV.
Methods: The study was conducted atDepartment of Obstetrics and Gynecology and Department of Paediatrics, secondary level referral centre, MCH block, District hospital, Vizianagaram, Andhra Pradesh, India. The data was collected during May2009 to May 2018 from records of maternal details registered at PPTCT clinic as well as list of infants undergoing Early Infant Diagnosis (EID) recorded in the standard format as per instructions from National AIDS Control Organization (NACO) of India.The study was based on administration of Nevirapine drops and Cotrimoxazole syrup as per the guidelines of National AIDS Control Organization (NACO).
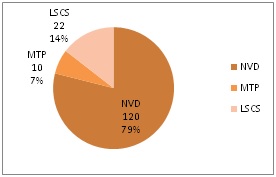
Results: A total of 68,000 babies were born during 2009 to 2018, of which 63320 were live born, among them 152 were born to HIV positive mothers. All the 152 babies were given Nevirapine drops immediately after delivery followed by cotrimoxazole syrup and tested for HIV antibodies at 6 months, 12 months and 18months of age. Amongst all the babies born to HIV positive mothers, only one baby was HIV reactive, and moreover no baby developed any life threatening adverse reaction in this study.
Conclusion:The follow up study of new born babies of HIV positive mothers on Nevirapine drops is found to be effective and may be continued as per the guidelines of NACO.
Downloads
References
2. Ramachandran R, Chandrasekaran V, Muniyandi M, Jaggarajamma K, Bagchi A, Sahu S. Prevalence and Risk Factors of HIV Infection among Clients Attending ICTCs in Six Districts of Tamilnadu, South India. HARTOJ. 2011; 2011: 1-7. doi:10.1155/2011/650321.
3. National AIDS Control Organization (NACO) releases HIV Estimations 2017. 14 SEP 2018 3:39PM; PIB Delhi, Ministry of Health and Family Welfare, Government of India.
4. The Working Group on Mother-to-Child Transmission of HIV. Rates of mother-to- child transmission of HIV-1 in Africa, America, and Europe: results from 13 perinatal studies. J Acquir Immune DeficSyndr Hum Retrovirol. 1995; 8(5):506–510
5. De Cock KM, Fowler MG, Mercier E, de Vincenzi I, Saba J, Hoff E, Alnwick DJ, Rogers M, Shaffer N. Prevention of mother-to-child HIV transmission in resource poor countries: translating research into policy and practice. JAMA. 2000; 283(9):1175–1182.[pubmed]
6. Children with HIV and AIDS [Internet]. 2018 [cited 2018 Dec 14]. Available from: https://www.webmd.com /hiv-aids/ guide/hiv-in-children#1-2.
7. Chukwuemeka IK, Fatima MI, Ovavi ZK, Olukayode O. The impact of a HIV prevention of mother to child transmission program in a Nigerian early infant diagnosis centre. Niger Med J. 2014; 55(3):204-08.
8. WHO. Consolidated Guidelines on the use of antiretroviral drugs for treating and preventing HIV Infection: recommendations for a public health approach. Geneva: WHO Press 2013. Available from: http://apps. who.int/iris/ bitstream/ 10665/85321 /1/9789241505727_eng.pdfua=1.
9. Pediatric Antiretroviral Therapy (ART)Antiretroviral Therapy (ART) Guidelines [Internet]. 2014. Available from: http://naco.gov.in/sites/default/files/Pediatric_14-03-2014.pdf.
10. National AIDS Control Organization (NACO): Annual Report 2016-2017. Ministry of Health and female welfare, India. 24: 355-403; Available from: http://www.naco.gov.in.
11. Robert MK, Bonita MDS, Joseph SG, Nina FS, Richard EB. Nelson text book of paediatrics, 1 – Volume. Philadelphia, United States. Elsevier - Health Sciences Division. 2011.
12. The Working Group on Mother-to-Child Transmission of HIV. Rates of mother-to-child transmission of HIV-1 in Africa, America, and Europe: results from 13 perinatal studies. J Acquir Immune DeficSyndr Hum Retrovirol. 1995;8(5):506–510pmid:7697448.
13. Perre PV de, Simonon A, Msellati P, Hitimana DG, Vaira D, Bazubagira A, Goethem CV, Stevens AM, Karita E, Thull DS, Dabis F, Lepage P. Postnatal transmission of human immunodeficiency virus type 1 from mother to infant - A prospective cohort study inKigali, Rwanda. N Engl J Med. 1991;325(9): 593–598.
14. Dunn DT, Newell ML, Ades AE, Peckham CS. Risk of human immunodeficiency virus type 1 transmission through breastfeeding. Lancet. 1992 Sep 5; 340(8819):585–588.[pubmed]
15. Nduati R, John G, Mbori-Ngacha D, Richardson B, Overbaugh J, Mwatha A, Ndinya-Achola J, Bwayo J, Onyango FE, Hughes J, Kreiss J. Effect of breastfeeding and formula feeding on transmission of HIV-1: a randomized clinical trial. JAMA. 2000 Mar 1; 283(9):1167–1174.[pubmed]
16. Dunn DT, Tess BH, Rodrigues LC, Ades AE. Mother-to-child transmission of HIV: implications of variation in maternal infectivity. AIDS. 1998; 12(16):2211–2216.[pubmed]
17. Committee on pediatric AIDs. Policy statement: Infant feeding and transmission of human immuno deficiency virus in the United States.Pediatrics. 2013 Feb;131(2): 391-396.
18. MofensonLM. Antiretroviral drugs to prevent breastfeeding HIV transmission.AntivirTher. 2010;15(4):537–553.doi: 10.3851/IMP1574.[pubmed]
19. Chintu C, Bhat GJ, Walker AS, Mulenga V, Sinyinza F, Lishimpi K, Farrelly L, Kaganson N, Zumla A, Gillespie SH, Nunn AJ, Gibb DM; CHAP trial team. Co-trimoxazole as prophylaxis against opportunistic infections in HIV-infected Zambian children (CHAP): a double-blind randomised placebo-controlled trial. Lancet. 2004 Nov 20-26; 364(9448):1865-71.[pubmed]
20. WHO issues guidelines on use of Co-trimoxazole prophylaxis [Internet]. 2006. Available from: http://www.aidsmap.com/WHO-issues-guidelines-on-use-of-cotrimoxazoleprophylaxis/page/1424561/.
21. Co-trimoxazole prophylaxis for HIV-exposedandHIV-infected infants and children: Practical approaches to implementation and scale up. Geneva: WHO Press 2009.Available from: https://www.unicef.org/ aids/files/CotrimoxazoleGuide_2009.pdf.
Copyright (c) 2019 Author (s). Published by Siddharth Health Research and Social Welfare Society

This work is licensed under a Creative Commons Attribution 4.0 International License.


 OAI - Open Archives Initiative
OAI - Open Archives Initiative


